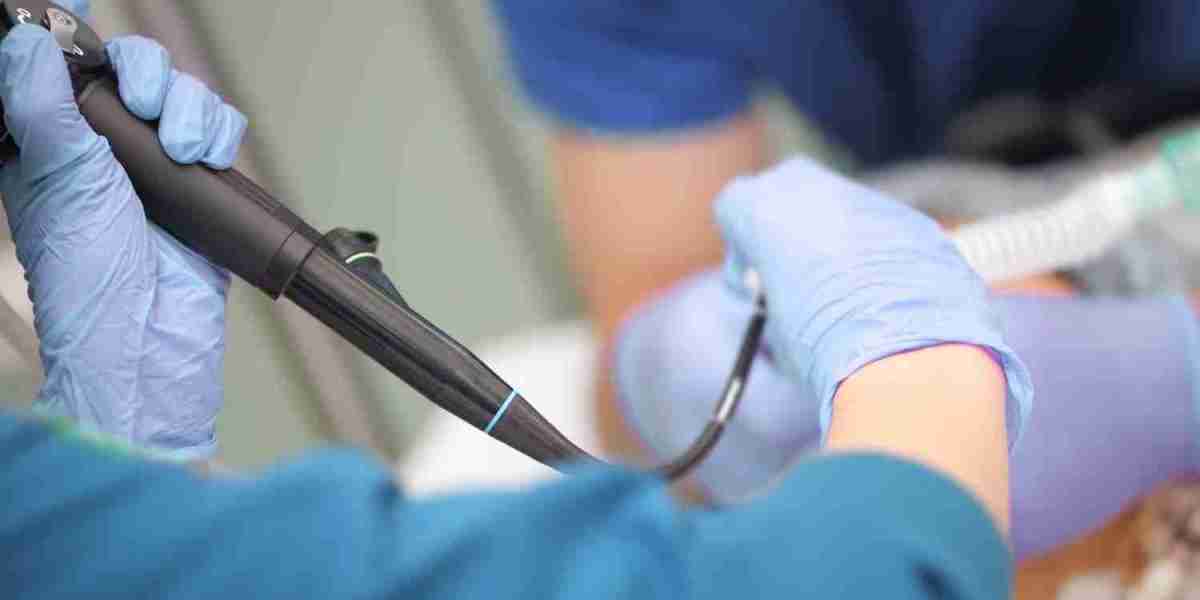
Introduction
The automated endoscope reprocessing market is segmented based on various factors, including product types, end-user industries, and geographic regions. Among these, product segmentation plays a crucial role in determining the market landscape, as different healthcare facilities require distinct reprocessing solutions. This article explores the market segmentation and key product types driving growth in the automated endoscope reprocessing sector.
Market Segmentation of Automated Endoscope Reprocessing
The automated endoscope reprocessing market can be segmented into the following categories:
1. By Product Type
Single-Basin Automated Endoscope Reprocessors (AERs):
Designed for lower-volume healthcare facilities.
Cost-effective and compact.
Suitable for small clinics and outpatient centers.
Dual-Basin Automated Endoscope Reprocessors (AERs):
Provide higher throughput by processing multiple endoscopes simultaneously.
Ideal for hospitals and large healthcare centers with high patient volumes.
Offer greater efficiency in infection control management.
Endoscope Drying and Storage Systems:
Ensure proper drying and storage to prevent microbial contamination.
Maintain sterility and prolong the life of reprocessed endoscopes.
Increasingly adopted due to stringent infection control regulations.
2. By End-user Industry
Hospitals:
Largest segment in the market due to high patient volume.
Require high-capacity AERs and integrated drying/storage solutions.
Strong focus on compliance with regulatory guidelines.
Clinics and Ambulatory Surgical Centers (ASCs):
Growing adoption of compact and cost-efficient reprocessing units.
Preference for single-basin AERs with automated disinfection cycles.
Increasing regulatory pressure driving investments in better reprocessing solutions.
3. By Region
North America:
Leading market due to advanced healthcare infrastructure.
Strict infection control regulations driving demand for AERs.
Presence of key industry players accelerating technological innovation.
Europe:
Strong emphasis on sustainability and eco-friendly reprocessing solutions.
Compliance with EU sterilization and medical device standards.
Increasing adoption of AI-driven reprocessing systems.
Asia-Pacific:
Rapid healthcare expansion in emerging economies.
Growing awareness about infection control measures in hospitals and clinics.
Rising investments in healthcare infrastructure fueling demand for automated solutions.
Key Product Types in Automated Endoscope Reprocessing
1. High-Level Disinfection (HLD) Systems
Ensure complete elimination of microbial contamination.
Use of chemical disinfectants such as peracetic acid and hydrogen peroxide.
Widely used in hospitals and specialized endoscopy centers.
2. Smart and AI-powered Reprocessing Units
Integration of AI-driven tracking and automation to optimize reprocessing cycles.
Automated compliance reporting to meet regulatory standards.
Reduce operational costs by improving resource efficiency.
3. Portable and Compact AERs
Designed for small healthcare settings with limited space.
Enable point-of-care reprocessing, reducing workflow interruptions.
Cost-effective solutions catering to ambulatory surgical centers and clinics.
Conclusion
The automated endoscope reprocessing market is characterized by diverse product offerings tailored to different healthcare needs. From high-capacity dual-basin AERs used in hospitals to portable single-basin units preferred by clinics, the market continues to expand with technological advancements and regulatory-driven innovations. As demand for infection control intensifies, automated endoscope reprocessing solutions will play a pivotal role in ensuring patient safety and operational efficiency across healthcare facilities.

![Dofus Quêtes - Le disparu de Sufokia [Guide]](https://pungi.b-cdn.net/upload/photos/2025/03/4nQqxE7rwlPRK1LE2NaY_14_e8a971224d326154020d28a2b9666934_image.png)
